37 the ideal gas law worksheet answers
The ideal and combined gas laws worksheet answers chemfiesta Created Date. We can calculate the volume of 1000 mol of an ideal gas under standard conditions using the variant of the ideal gas law given in Equation 1047. Ideal Gas Law Practice Worksheet Solve the following problems using the ideal gas law. Kmol If pressure is needed in kPa then ...
The Ideal Gas Law Worksheet Answers 1 Small Children Are Occasionally Injured When They Try To Inhale Helium From A Compressed Helium Tank. N PV 28 atm98 L 11 moles of gas RT 00821 LatmmolK292 K 2 If 50 moles of O 2 and 30 moles of N 2 are placed in a 300 L tank at a temperature of 25 0. A container containing 500 l of a gas is collected at.
Key ideal gas law worksheet answers. The value of r varies with the units chosen. A sample of hydrogen gas has a volume of 8 56 l at a temperature of 0 o c and a pressure of 1 5 atm. K mol if pressure is needed in kpa then convert by multiplying by 101 3kpa 1atm to get r 8 31 kpa l k mole 1 if i have 4 moles of a gas at a pressure of 5 6 atm ...

The ideal gas law worksheet answers
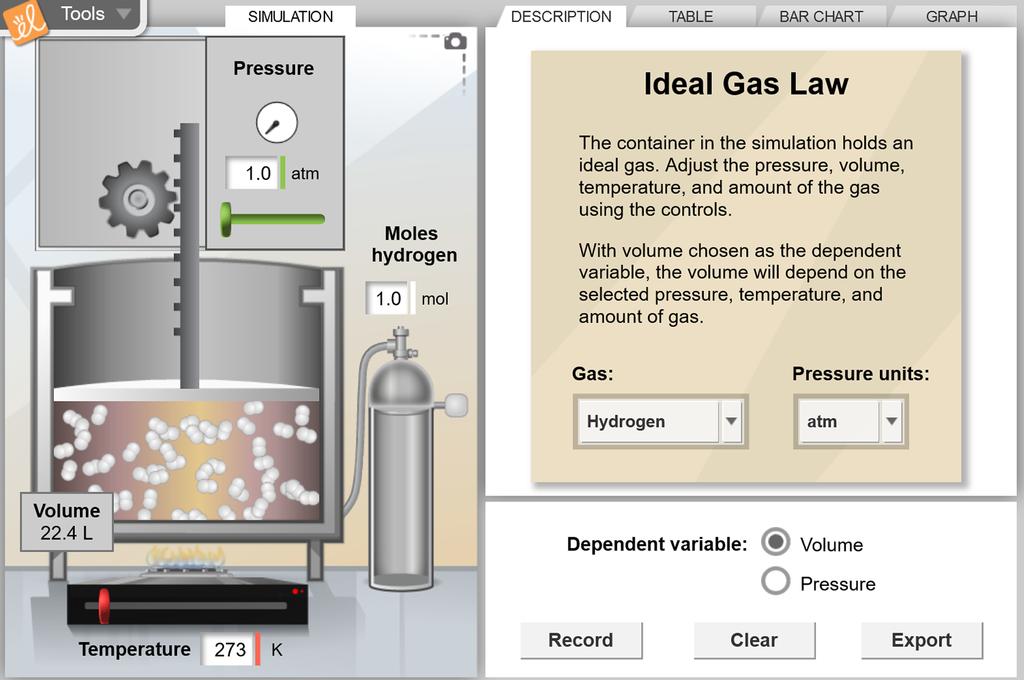
Worksheet 7 - Ideal Gas Law I. Ideal Gas Law The findings of 19th century chemists and physicists, among them Avogadro, Gay-Lussac, Boyle and Charles, are summarized in the Ideal Gas Law: PV = nRT P = pressure V = volume n= moles of gas, R = universal gas constant T = temperature. The value of R varies with the units chosen: R = 0.08206 L atm / mol K
Gas Law Worksheet Answer Details. 1. A cylinder of argon gas contains 50.0 L of Ar at 18.4 atm and 127 °C. How many moles of argon are in the cylinder?3 pages
Solve the following problems using the ideal gas law: 1). How many moles of gas does it take to occupy 120 liters at a pressure of. 2.3 atmospheres and a ...2 pages
The ideal gas law worksheet answers.
2) At what temperature would 2.10 moles of N2 gas have a pressure of 1.25 atm and in a 25.0 L tank? 3) When filling a weather balloon with gas you have to consider that the gas will expand greatly as it rises and the pressure decreases. Let's say you put about 10.0 moles of He gas into a balloon that can inflate to hold 5000.0L. Currently,
Problem #9a: What is the value of and units on R? What is R called ("A letter" is not the correct answer!)? R is called the gas constant. It was first ...
Ideal Gas Law Worksheet PV = nRT Ideal Gas Law Worksheet PV = nRT Use the ideal gas law, "PerV-nRT", and the universal gas constantR = 0.0821 L*atm to solve the following problems:K*mol If pressure is needed in kPa then convert by multiplying by 101.3kPa / 1atmto get R =8.31 kPa*L / (K*mole)
Ideal Gas Law Gizmo Worksheet Answers. Wallpaper Cosmic Worksheet September 4, 2021. K mol if pressure is needed in kpa then convert by multiplying by 101 3kpa 1atm to get r 8 31 kpa l k mole 1 if i have 4 moles of a gas at a pressure of 5 6 atm and a volume of 12. On the boyle s law tab set the temperature to 300 k and press ok.
CH301 Worksheet 8—Gases (Answer Key) 1. What do we assume about ideal gases? What is the ideal gas law? Give the units for each variable. Ideal gases are infinitely small, hard spheres that do not interact with each other. They are essentially "blind" to other gas molecules and will bounce off of each other just as they would bounce of a wall.
Gas Laws Worksheet Answer Key -. The ideal gas law states that pv nrt where p is the pressure of a gas v is the volume of the gas n is the number of moles of gas present r is the ideal gas constant and t is the temperature of the gas in kelvins. The tire volume can only expand so much earlier the rubber dà and releases the accumulation.
If a gas is cooled from 343.0 K to 283.15 K and the volume is kept constant what final pressure would result if the original pressure was 760.0 mm Hg? Ideal Gas Law Problems: PV =nRT. R = 0.0821 L*atm P is in atm T is in Kelvin V is in Liters. K*mol
The ideal gas law states that pv nrt where p is the pressure of a gas v is the volume of the gas n is the number of moles of gas present r is the ideal gas constant and t is the temperature of the gas in kelvins. Ideal gas law practice worksheet solve the following problems using the ideal gas law.
Afrikaans Grade 7 Exam Papers. Displaying top 8 worksheets found for - Afrikaans Grade 7 Exam Papers. Some of the worksheets for this concept are Grades 4, Exams, English comprehension and language grade 7 2011, Tom newby school examination, Friday 10 february afrikaans gr 7 eng gr 4 6, Grade 7 practice test, Creative arts drama assessment task november 2014 grade 8, June exams 2018 grade 8 ...
Write your answer on the line provided. _____ 1. Real gases behave like ideal gases except at very high temperatures. _____ 2. The gas constant, R, is equal to 0.0821 when the pressure is expressed in . kilopascals. _____ 3. As more gas particles are added to a container, there are fewer collisions
The constant R in this equation is known as the universal gas constant Chemistry the ideal gas law worksheet answers. It arises from a combination of the proportionality constants in the three empirical gas laws. The universal gas . . Chemistry the ideal gas law worksheet answers. Go to the Ideal Gas Worksheet. Go to the Gas Law Review.
Ideal gas law worksheet answers. The ideal gas law is ideal because it ignores interactions between the gas particles in order to simplify the equation. Calculate the moles of hydrogen present in the sample. Solutions to the ideal gas law practice worksheet. How would this answer change if the gas had been helium.
The ideal gas law states that PV=nRT, where P is the pressure of a gas, V is the volume of the gas, n is the number of moles of gas present, R is the ideal gas constant, and T is the temperature of the gas in Kelvins. Common mistakes: Make sure you T in Kelvins, rather than degrees celsius.
Examples & practice on ideal gas law worksheet! Stoichiometry of Gases Can do L-L conversions (just like mol-mol) with 2 gases and an equation Example: Given: C 3H 8 (g) + 5 O 2 (g) à 3 CO 2 (g) + 4 H 2O (l) How many L of O 2 are required to react with 0.35 L of propane? **You may have to use molar volume!** **You may have to use the ideal gas ...
Ideal gas laws worksheet & Ideal Gas Law Worksheet Answers from Ideal Gas Law Worksheet Answers, source: ngosaveh.com. Learn quiz on ideal gas constant chemistry quiz 111 to practice from Ideal Gas Law Worksheet Answers, source: pinterest.co.uk. 30 Inspirational Ideal Gas Law Worksheet from Ideal Gas Law Worksheet Answers, source: coletivocompa.org
Transcribed image text: Experiment 14: Using the Ideal Gas Law to Measure Gas Produced from a Reaction PREPARATION WORKSHEET Name: Date: TA: Section: Answer the following questions using information obtained from lecture, the textbook, the lab manual, or other reliable sources. The responses will be collected at the beginning of Pre-Lab Recitation and checked at the start of your lab section ...
Avogadro's Law - relationship between moles and volume . A sample of hydrogen gas has a volume of 8.56 L at a temperature of 0 o C and a pressure of 1.5 atm. Calculate the moles of hydrogen present in the sample. 0.57 moles . How would this answer change if the gas had been helium? There would be no difference in the answer.
This is a single 2-page worksheet with problems utilizing the ideal gas law. Students will solve for each of the variables, and for molar mass. There are a total of 8 problems.Answer key is included.The download includes a handout master (.pdf) that includes one worksheet, and answer key.This produc. Subjects:
The Ideal and Combined Gas Laws PV = nRT or P1V1 = P2V2 T 1 T2 The Ideal and Combined Gas Laws PV = nRT or P 1V 1= P 2V 2 T 1T 2 Use your knowledge of the ideal and combined gas laws to solve the following problems. If it involves moles or grams, it must be PV = nRT
Key ideal gas law worksheet answers. A sample of hydrogen gas has a volume of 8 56 l at a temperature of 0 o c and a pressure of 1 5 atm. Calculate the moles of hydrogen present in the sample. Ideal gas law the findings of 19th century chemists and physicists among them avogadro gay lussac boyle and charles are summarized in the ideal gas law.
Ideal Gas Law Worksheet PV = nRT Use the ideal gas law, "PerV-nRT", and the universal gas constant R = 0.0821 L*atm to solve the following problems: K*mol If pressure is needed in kPa then convert by multiplying by 101.3kPa / 1atm to get R =8.31 kPa*L / (K*mole) Ideal Gas Law Chemistry Test Questions - ThoughtCo
Jun 17, 2021 · Learning Objectives. To recognize the differences between the behavior of an ideal gas and a real gas; To understand how molecular volumes and intermolecular attractions cause the properties of real gases to deviate from those predicted by the ideal gas law.
Key ideal gas law worksheet answers. Ideal gas law the findings of 19th century chemists and physicists among them avogadro gay lussac boyle and charles are summarized in the ideal gas law. If the pressure exerted by a gas at 25 c in a volume of 0 044 liter is 3 81 atm how many moles of. The value of r varies with the units chosen.
The ideal gas law states that pv nrt where p is the pressure of a gas v is the volume of the gas n is the number of moles of gas present r is the ideal gas constant and t is the temperature of the gas in kelvins. P 1 v 1 n 1 rt 1. Charles Law Chemistry Notes Gas Laws Chemistry Chemistry 42 Math Conversion Worksheets For Grade 7 Di 2020
Moore at www.learning.mgccc.cc.ms.us/pk/sciencedocs/gaslawwksheet.htm Directions: Answer each question below. Then write the name of the gas law used to solve ...
In addition mass and molecular weight will give us moles. Ideal gas law practice problems worksheet answers. Solutions to the ideal gas law practice worksheet. Using the ideal gas law to calculate number of moles. 1 2 3 moles of helium gas are at a pressure of 1 70 atm and the temperature is 41 c. 1120 torr 1120 mm hg 149 kpa 2.
Ideal gas law practice worksheet with answers Problem (PageIndex {1}) Sometimes leaving a bicycle under the sun on a hot day causes a burst. Why? Answer as a temperature of a gas increases, the pressure will also increase according to the ideal gas law. The tire volume can only expand so much earlier the rubber dà and releases the accumulation ...
Use the Ideal Gas Law below to solve the following problems. PV = nRT where P = pressure in atmospheres. V = volume in liters n = number of moles of gas.1 page
What is the gas? Answers: 1. 60.0 L o2. 59 g CO 3. 517.6 kPa 4. -112 C 5. radon . Chemistry: The Ideal Gas Law KEY Directions: Solve each of the following problems. Show your work, including proper units, to earn full credit. 1. If 3.7 moles of ...
Collection Of Gas Law Activities Ideal Gas Law Cooperative Learning Activities Activities. Chemical Bonding Crossword Worksheet Answers Worksheets Are A Crucial Part Of Studying English Todd In 2021 Puzzles And Answers High School Science Crossword Puzzle. Ideal Gas Law Practice Problems Ideal Gas Law Chemistry Lecture Lectures Notes.































0 Response to "37 the ideal gas law worksheet answers"
Post a Comment